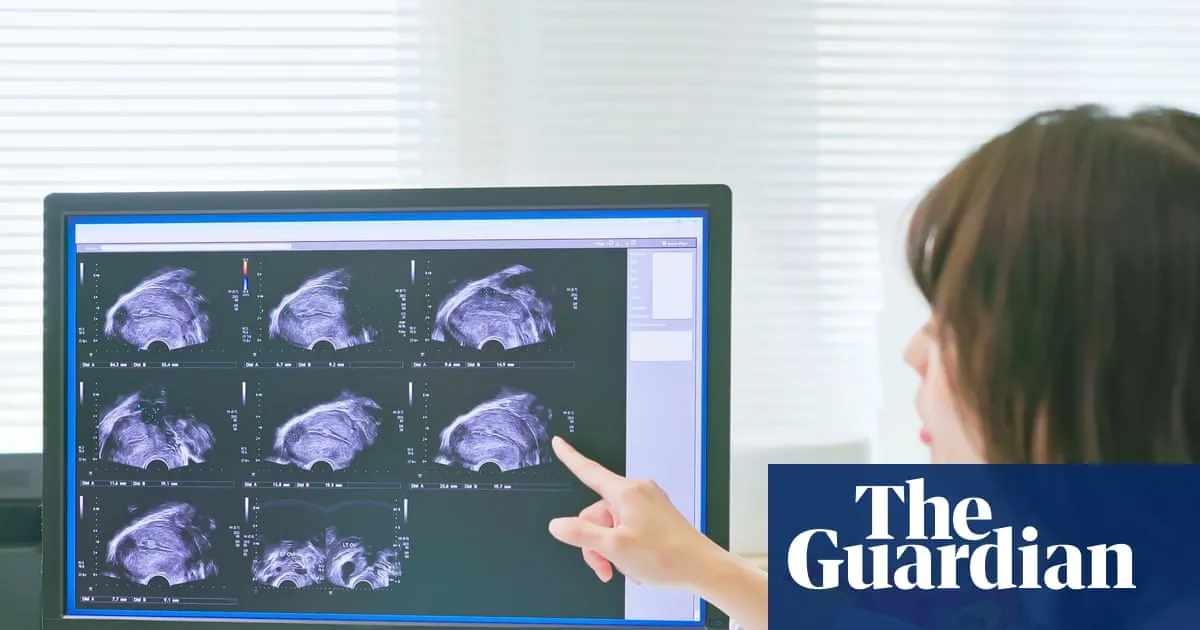
An experimental scanning technique for endometriosis has produced encouraging results in an early clinical trial, raising hopes that the condition could one day be diagnosed without the need for invasive surgery, according to findings reported by the Guardian.
The trial, which involved 19 women diagnosed with endometriosis, tested a radiotracer compound called maraciclatide. Researchers found that the substance was able to highlight endometriosis lesions on a scan, effectively making the condition visible in a way that conventional imaging has not reliably achieved.
Why diagnosis has been difficult
Endometriosis affects tissue that resembles the lining of the uterus, which grows outside of it and can cause chronic pain, fertility problems, and other serious complications. Despite its prevalence, the condition is notoriously difficult to diagnose without surgical intervention.
At present, a definitive diagnosis typically requires a laparoscopy - a procedure in which a surgeon inserts a small camera into the abdomen to examine the pelvic organs directly. This surgical requirement is widely regarded as a significant barrier to timely diagnosis, contributing to delays that can stretch for years in many cases.

How the radiotracer works
Maraciclatide is designed to bind to specific proteins present in endometriosis tissue. When introduced into the body ahead of a scan, it effectively causes affected areas to appear prominently on imaging, allowing clinicians to identify the condition without opening the body surgically.
The trial results suggest this approach has potential as a non-invasive diagnostic pathway, though researchers cautioned that the study was small in scale and that further, larger trials would be needed before any clinical application could be considered.
Significance for patients
For patients, the prospect of a scan-based diagnosis carries considerable significance. The current diagnostic journey is frequently lengthy and distressing, with many women reporting that their symptoms are dismissed or misattributed before a confirmed diagnosis is reached.
A reliable non-invasive test could reduce the time between the onset of symptoms and confirmed diagnosis, potentially allowing earlier access to treatment and better long-term health outcomes.
Researchers involved in the work described the findings as a meaningful step forward, while emphasising that the trial was an early-stage proof-of-concept study. Further clinical investigation will be required to establish the safety, accuracy, and practicality of the approach at scale.